 
3 rd orbit (energy level) is represented as M shell and it can contain up to 18 electrons.2 nd orbit (energy level) is represented as L shell and it can hold up to 8 electrons.1 st orbit (energy level) is represented as K shell and it can hold up to 2 electrons.However, when an electron loses energy it moves from higher to lower energy level. In an atom, the electrons move from lower to higher energy level by acquiring the required energy. The change in energy occurs when the electrons jump from one energy level to other.Learn the concept of Valency here in detail here. The lowest energy level of the electron is called the ground state. The different energy levels or orbits are represented in two ways such as 1, 2, 3, 4… or K, L, M, N…. 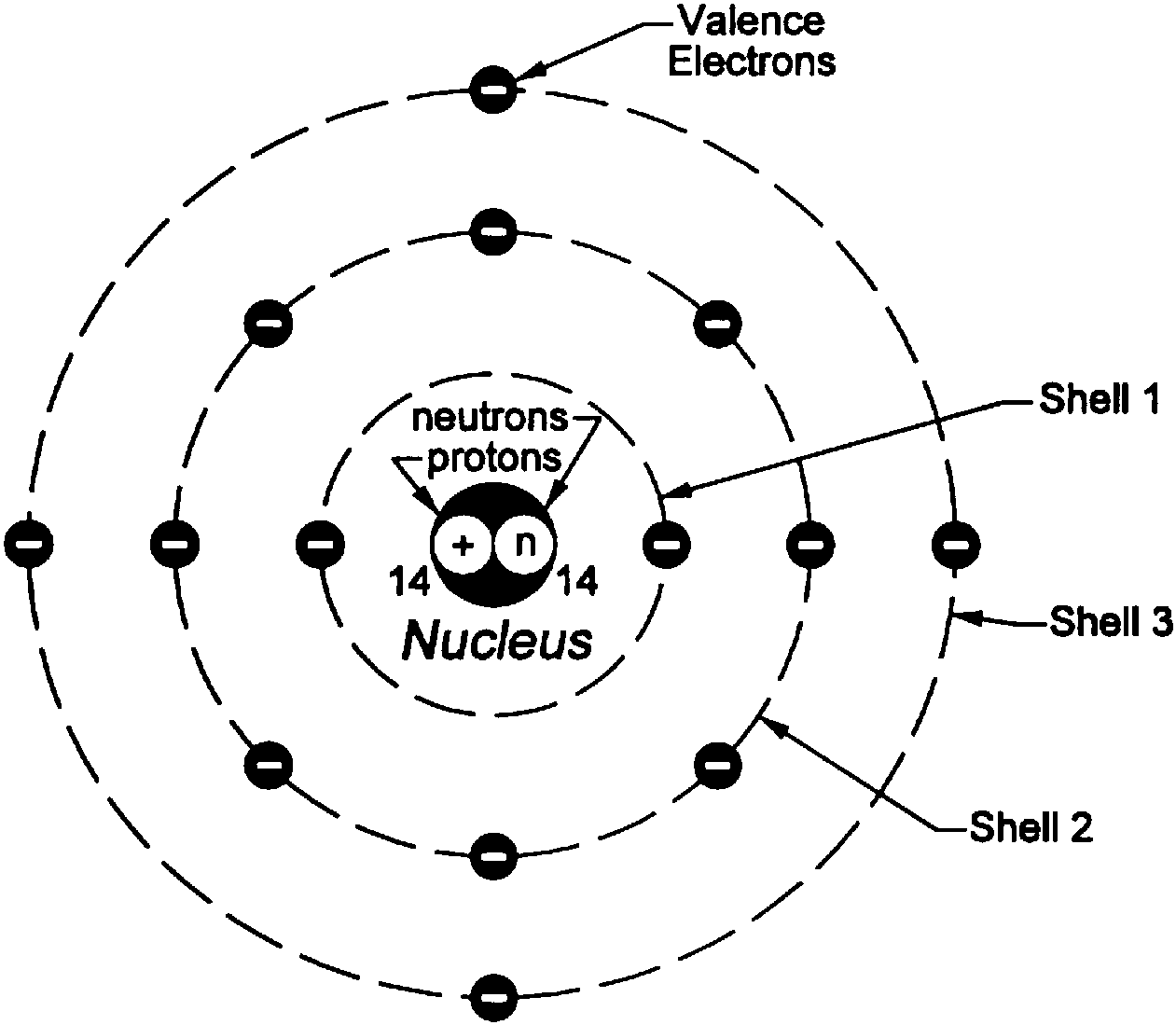
Learn the concept of an Atomic number here. 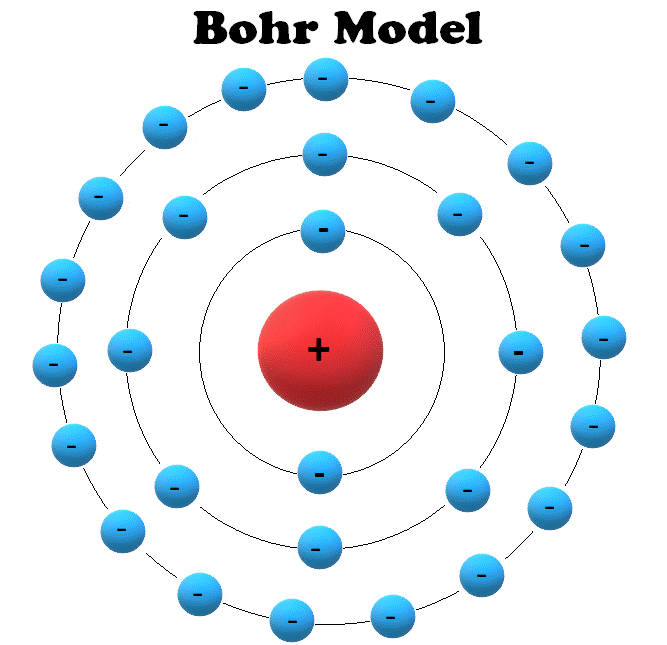
The range of quantum numbers may vary and begin from the lowest energy level (nucleus side n=1) to the highest energy level.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
